Peptide reconstitution lab protocol: a precise guide
Errors in peptide reconstitution are among the most common and least discussed sources of experimental failure in biomedical research. A lyophilized peptide vial that looks intact can yield a compromised solution if the reconstitution steps are rushed, performed without proper materials, or executed without attention to technique. Following a validated peptide reconstitution laboratory protocol is not a formality — it is the difference between reproducible data and results that cannot be trusted. This guide covers every stage: material preparation, execution, concentration calculation, storage, and the errors that silently undermine research quality.
Table of Contents
- Materials and preparation essentials for peptide reconstitution
- Step-by-step peptide reconstitution procedure
- Calculating concentration and ensuring dosing accuracy
- Storage and handling guidelines for reconstituted peptides
- Common errors in peptide reconstitution and how to avoid them
- Why precision and patience matter beyond the protocol
- Discover how Vertex Labs supports your peptide reconstitution accuracy
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Use bacteriostatic water | Bacteriostatic water with benzyl alcohol preserves peptide solutions better than sterile water, extending shelf life during multi-dose use. |
| Add solvent slowly | Inject solvent gently down the vial wall to avoid peptide denaturation caused by mechanical stress and direct impact. |
| Avoid shaking peptides | Gently swirl or roll the vial after solvent addition to dissolve peptides without introducing damaging air bubbles. |
| Calculate concentration precisely | Accurate dose calculations depend on correct solvent volume and peptide mass to maintain experimental consistency. |
| Store vials properly | Keep reconstituted peptides refrigerated at 2–8°C, labeled clearly, and use them within the recommended timeframe to maintain integrity. |
Materials and preparation essentials for peptide reconstitution
A sound peptide reconstitution laboratory protocol begins well before the syringe is loaded. Research-grade peptides are supplied as lyophilized powder and must be reconstituted carefully using specific materials to ensure stability and sterility. Gathering everything before you open a vial eliminates mid-process improvisation, which is a primary cause of contamination and dosing error.
Essential materials checklist:
- Lyophilized peptide vial (verified by Certificate of Analysis)
- Bacteriostatic water (BAC water) as the preferred reconstitution solvent
- 1–3 mL syringe with an 18–23 gauge needle for solvent transfer
- U-100 insulin syringe for dose measurement (separate from reconstitution)
- Alcohol prep wipes (70% isopropyl)
- Sterile field or laminar flow hood if available
- Permanent marker and labels for immediate vial identification
Why bacteriostatic water over sterile water? Bacteriostatic water contains 0.9% benzyl alcohol, which inhibits bacterial growth in multi-dose vials. Sterile water lacks this preservative, making it appropriate only for single-use reconstitution. For most research applications where a vial will be accessed multiple times, BAC water extends usable solution life considerably. Refer to peptide reconstitution best practices for solvent selection guidance specific to peptide class.
Allow the lyophilized vial to equilibrate at room temperature for 15 to 20 minutes before opening. This step is frequently skipped, but it matters. A cold vial exposed to ambient air develops condensation on the stopper and interior surfaces, which can dilute the peptide or introduce moisture before you add your measured solvent volume. Equilibration prevents this.
Pro Tip: Swab the rubber stopper with an alcohol wipe and allow it to dry completely for at least 30 seconds before needle insertion. A wet stopper can introduce isopropyl alcohol into the vial, which may affect peptide stability.
| Material | Specification | Purpose |
|---|---|---|
| Bacteriostatic water | USP grade, 0.9% benzyl alcohol | Reconstitution solvent, multi-dose preservation |
| Reconstitution syringe | 1–3 mL, 18–23 gauge | Controlled solvent transfer |
| Insulin syringe | U-100, 0.3–1 mL | Precise dose measurement |
| Alcohol wipes | 70% isopropyl | Stopper and surface sanitization |
| Vial labels | Permanent, waterproof | Traceability and compliance |
With materials ready and sterile conditions established, you can move on to the precise reconstitution process.
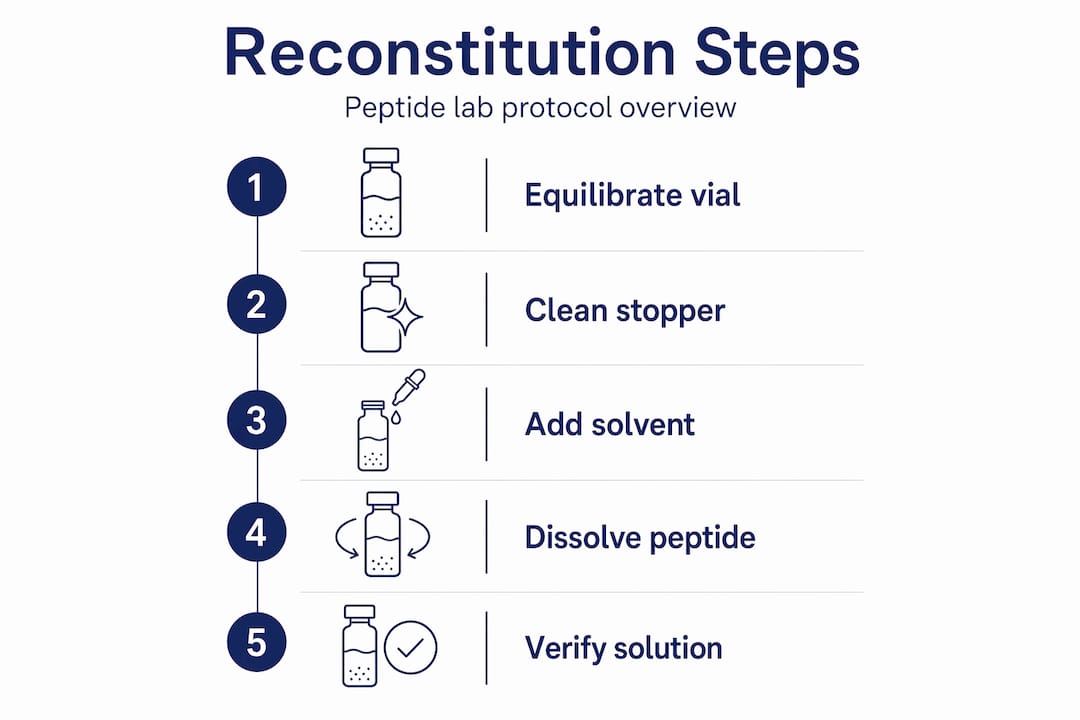
Step-by-step peptide reconstitution procedure
This is the stage where most errors occur. The following numbered protocol reflects standard laboratory peptide handling practices and includes the rationale for each step so you can adapt intelligently when working with less common peptide sequences.
- Confirm vial identity and COA. Before touching the vial, verify the peptide name, batch number, and purity against the Certificate of Analysis. Never reconstitute an unlabeled or unverified vial.
- Allow vial to reach room temperature. Wait 15–20 minutes after removing from cold storage. Do not accelerate this with heat.
- Prepare your sterile workspace. Wipe down the work surface with 70% isopropyl alcohol. If a laminar flow hood is available, use it.
- Swab and dry the rubber stopper. Apply an alcohol wipe to the stopper and allow it to air-dry for 30 seconds minimum.
- Draw the calculated solvent volume into a 1–3 mL syringe. Do not use an insulin syringe for this step; the small barrel volume makes controlled injection difficult and increases the risk of pressure-related peptide damage.
- Insert the needle at a 45° angle and direct the solvent stream slowly down the inner wall of the vial. Adding solvent slowly down the vial wall prevents mechanical denaturation and reduces bubbles; shaking causes peptide aggregation and loss of activity.
- Do not inject directly onto the peptide cake. Directing solvent forcefully onto the lyophilized powder disrupts the cake structure and causes clumping that resists dissolution.
- Gently swirl or roll the vial between your palms after injection. Avoid vigorous shaking; gentle swirling or rolling is recommended to dissolve the powder without compromising peptide integrity.
- If the peptide does not dissolve fully, refrigerate the vial for 5 minutes and swirl again. Mild sonication in a water bath sonicator is acceptable as a last resort for difficult sequences, but it should not be the default approach.
- Inspect the solution. A properly reconstituted peptide solution should be clear or very slightly opalescent. Visible particulates or cloudiness may indicate incomplete dissolution or degradation.
“The reconstitution step is not mechanical — it is a controlled dissolution process. The physical forces you apply directly affect the bioactivity of the final solution.”
Pro Tip: For hydrophobic peptides that resist aqueous dissolution, add a small volume (5–10%) of glacial acetic acid or DMSO before adding BAC water. This pre-wets the peptide cake and significantly improves dissolution without compromising most research applications.
After careful reconstitution, it is essential to verify your solution’s quality and properly calculate concentrations before use.

Calculating concentration and ensuring dosing accuracy
Concentration errors are the second most common source of experimental failure in peptide research, after reconstitution technique errors. The math is straightforward, but the consequences of getting it wrong are significant: overdosing wastes material and skews data; underdosing produces null results that are misattributed to the peptide rather than the protocol.
Core formula:
Concentration (mcg/mL) = Total peptide mass (mcg) ÷ Solvent volume added (mL)
For example: a 5 mg (5,000 mcg) peptide vial reconstituted with 2.5 mL of BAC water yields a concentration of 2,000 mcg/mL.
Precise volume and concentration calculations are critical to avoid dosing errors. Once concentration is established, dose volume is calculated as:
Dose volume (mL) = Desired dose (mcg) ÷ Concentration (mcg/mL)
Practical dosing accuracy guidelines:
- Choose a solvent volume that aligns syringe graduation marks with target doses. User-friendly solvent volumes align syringe marks with desired doses to minimize mental math and reduce dosing errors.
- Never interchange syringes marked in different unit systems (e.g., reading a U-40 syringe as if it were U-100).
- Use the 1–3 mL reconstitution syringe only for adding solvent. Switch to a U-100 insulin syringe for all dose draws.
- Document your concentration calculation in the experiment log at the time of reconstitution, not retroactively.
| Peptide mass | Solvent volume | Concentration | Dose (mcg) | Draw volume |
|---|---|---|---|---|
| 5,000 mcg | 2.5 mL | 2,000 mcg/mL | 200 mcg | 0.10 mL (10 IU) |
| 5,000 mcg | 5.0 mL | 1,000 mcg/mL | 200 mcg | 0.20 mL (20 IU) |
| 2,000 mcg | 2.0 mL | 1,000 mcg/mL | 100 mcg | 0.10 mL (10 IU) |
Pro Tip: When working with a U-100 insulin syringe, remember that 1 IU on the barrel equals 0.01 mL. At 1,000 mcg/mL concentration, every 10 IU mark equals exactly 100 mcg. Designing your solvent volume around this relationship eliminates fractional syringe readings and reduces dosing error significantly. See our peptide concentration calculation resources for additional worked examples.
With concentration calculated and solution verified, proper storage and handling ensure peptide stability during use.
Storage and handling guidelines for reconstituted peptides
Reconstitution does not end at dissolution. How you store and handle the vial afterward determines how long the peptide remains viable for research use. Reconstituted peptides should be stored at 2–8°C and used within approximately 30 days; avoid freeze-thaw cycles which cause denaturation.
Storage and handling best practices:
- Store unopened lyophilized vials at -20°C for long-term preservation. Many peptides remain stable for 12 to 24 months under these conditions.
- Once reconstituted, transfer vials to 2–8°C refrigeration. This is the working storage temperature, not -20°C.
- Do not freeze reconstituted solutions unless the peptide’s stability data specifically supports it. Freeze-thaw cycling introduces ice crystal formation that physically disrupts peptide structure.
- Wrap vials in aluminum foil or store in opaque containers to protect light-sensitive sequences from photodegradation.
- Minimize the time each vial spends at room temperature. Return it to the refrigerator promptly after each draw.
“Every unnecessary minute a reconstituted vial spends at room temperature is a reduction in usable shelf life. Temperature cycling is cumulative damage.”
Labeling each vial with date, concentration, and solvent type supports traceability and reproducibility in research. Label immediately after reconstitution, not after the experiment. An unlabeled vial in a shared lab refrigerator is a liability.
Pro Tip: Use pre-printed label templates that include fields for peptide name, batch number, reconstitution date, concentration, solvent, and researcher initials. This takes 60 seconds and prevents the kind of misidentification errors that invalidate entire experimental runs. Review our peptide storage protocols for cold-chain management guidance.
Following proper storage, it is important to recognize and troubleshoot common errors to maintain research reliability.
Common errors in peptide reconstitution and how to avoid them
Even experienced researchers make reconstitution errors when working under time pressure or with unfamiliar peptide sequences. The following are the most consequential mistakes we see in laboratory peptide handling, along with direct corrective actions.
Errors and corrections:
- Using an insulin syringe to add BAC water. Insulin syringes for solvent addition or shaking the vial causes dosing imprecision and peptide denaturation. Use a 1–3 mL syringe for reconstitution only.
- Shaking the vial after solvent addition. This introduces air-liquid interfaces that promote aggregation. Swirling is always the correct technique.
- Injecting solvent directly onto the peptide cake. This causes clumping and incomplete dissolution. Always direct the stream down the vial wall.
- Leaving reconstituted vials at room temperature. Reconstituted vials at room temperature too long accelerates peptide degradation beyond bacteriostatic protection.
- Reusing needles for repeated vial access. Each insertion carries contamination risk. Always use a fresh needle for each draw.
- Failing to label vials immediately. An unlabeled vial is an untracked variable. Label at the moment of reconstitution.
“Protocol errors in reconstitution are rarely dramatic. They are quiet, cumulative, and often undetected until data reproducibility fails across multiple experiments.”
Pro Tip: Keep a dedicated reconstitution log adjacent to your peptide storage area. Record each vial access with the date, volume drawn, and remaining estimated volume. This practice supports traceability and helps you identify patterns in data variability that correlate with specific vial handling events. For a structured reference, consult our common peptide reconstitution errors resource.
Why precision and patience matter beyond the protocol
Following a written protocol is necessary. It is not sufficient. The variable that most consistently separates reliable peptide research from compromised studies is operator discipline — the willingness to follow each step completely, in sequence, without shortcuts, even when time is limited.
Consider the solvent injection step. The protocol says to inject slowly down the vial wall. In practice, under pressure, researchers inject faster than they should. The peptide solution still looks normal. The data, however, shows unexpected variability that gets attributed to the peptide source or the assay rather than the reconstitution technique. This is the category of error that is most damaging precisely because it is invisible.
Patience in allowing full dissolution before drawing a dose is equally important. A partially dissolved peptide cake means the first draws from a vial are more concentrated than later draws. This concentration gradient within a single vial is a reproducibility problem that cannot be corrected after the fact.
Meticulous labeling and storage habits are not administrative tasks. They are scientific controls. A vial reconstituted three weeks ago with an unknown solvent volume is a variable, not a reagent. Every unlabeled vial represents a potential confound in your data.
We have observed that labs which invest in standardized reconstitution procedures, including written SOPs, dedicated materials, and mandatory logging, consistently produce more reproducible data and waste less material. The protocol is the floor, not the ceiling. Technique, discipline, and environmental control determine how close your results come to the actual biology. For additional guidance, explore our insights on peptide reconstitution to support your team’s standardization efforts.
Discover how Vertex Labs supports your peptide reconstitution accuracy
Accurate reconstitution starts with verified source material. When the peptide itself is of uncertain purity or batch consistency, no protocol can fully compensate.
At Vertex Labs, we supply research-grade synthetic peptides with purity verified above 99% through third-party testing, accompanied by full Certificates of Analysis for every batch. Each order includes detailed product documentation to support your reconstitution and storage procedures. Our team is available to assist with protocol troubleshooting, solvent selection, and concentration planning for specific research applications. We offer a cold-chain delivery system designed to maintain peptide integrity from our facility to your laboratory. For institutions requiring consistent supply at scale, we also provide wholesale procurement options. When your research demands reliability at every step, your peptide source matters as much as your technique.
Frequently asked questions
Why is bacteriostatic water preferred over sterile water for peptide reconstitution?
Bacteriostatic water contains 0.9% benzyl alcohol, which inhibits bacterial growth during repeated vial access, extending peptide solution stability up to approximately 28 days when refrigerated, unlike sterile water which lacks preservatives and is suitable only for single-use reconstitution.
What happens if you shake a peptide vial vigorously after adding solvent?
Shaking introduces air bubbles and causes peptide denaturation at air-liquid interfaces, reducing potency and producing inaccurate dose concentrations; gentle swirling is the correct technique.
How long can reconstituted peptides be stored safely in the refrigerator?
Most synthetic peptide solutions stored at 2–8°C remain stable for approximately 30 days when reconstituted in bacteriostatic water, though sensitive sequences may require shorter storage windows and should be checked against available stability data.
What syringe should be used to add solvent versus draw doses?
Use a 1–3 mL syringe with an appropriate gauge needle to add solvent during reconstitution, and a separate U-100 insulin syringe for measuring and drawing precise dose volumes to avoid unit confusion and handling errors.
Why is labeling reconstituted peptide vials immediately important?
Unlabeled reconstituted vials introduce untraceable variables into experiments; accurate labeling with concentration, date, and solvent type is essential for dosing accuracy, experimental reproducibility, and compliance with laboratory documentation standards.